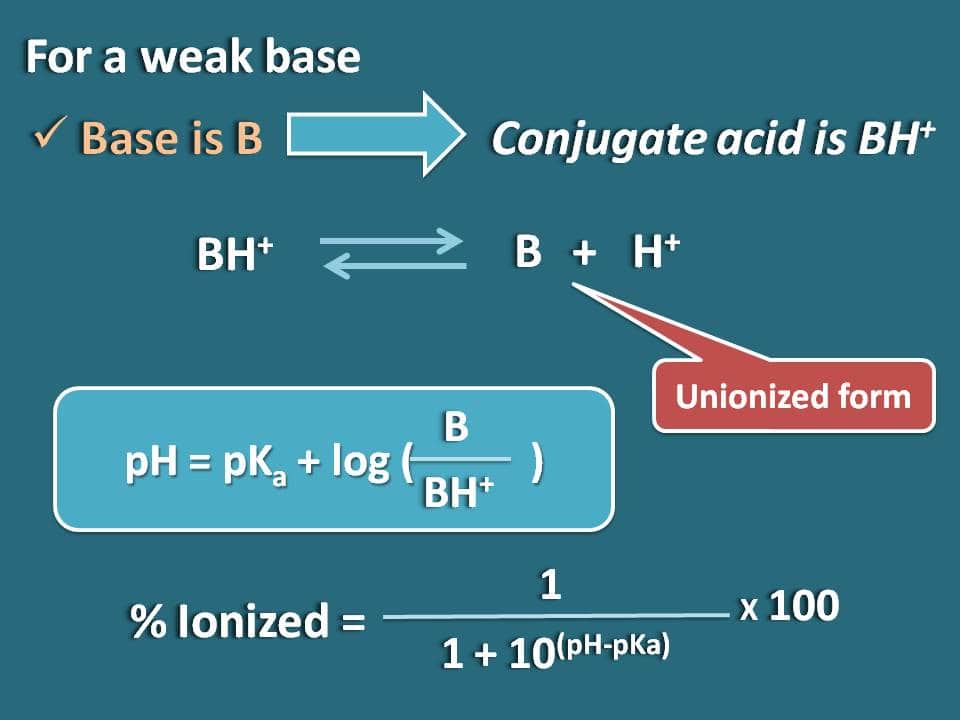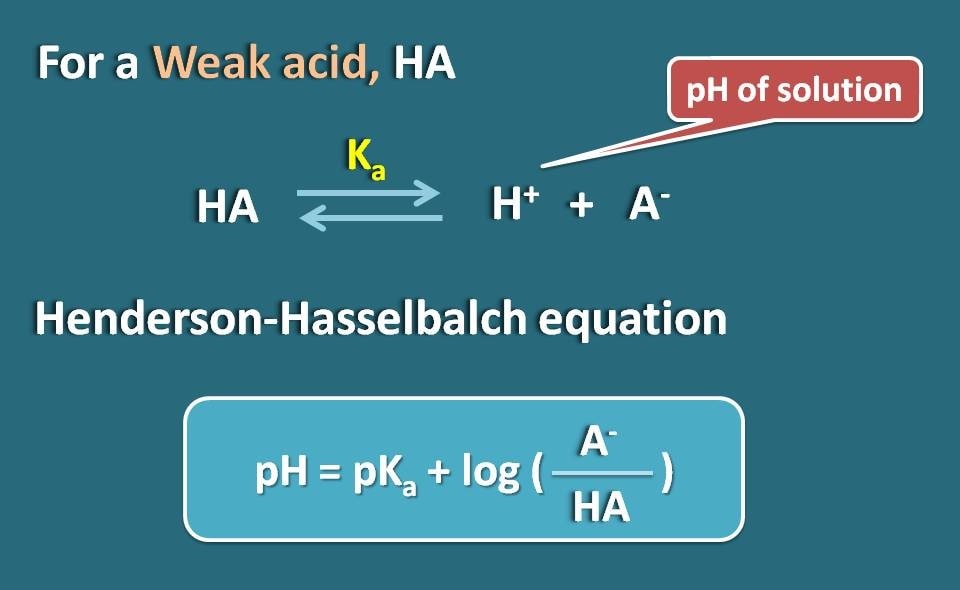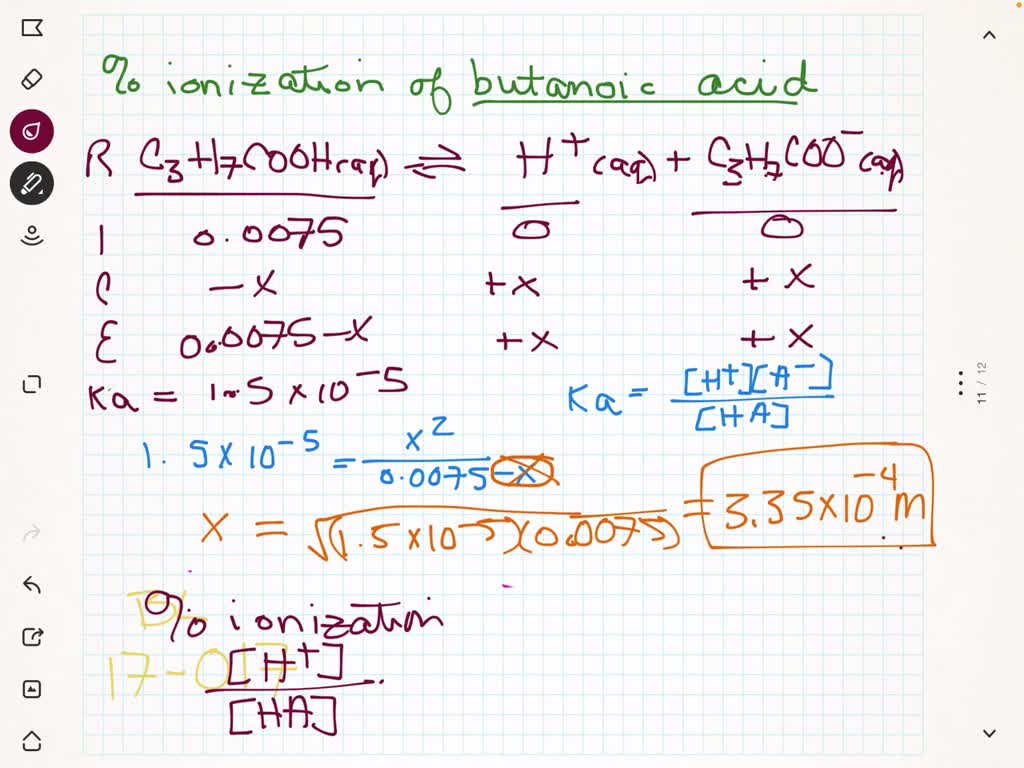
SOLVED:(a) Calculate the percent ionization of 0.0075 M butanoic acid (Ka=1.5 ×10^-5) . (b) Calculate the percent ionization of 0.0075 M butanoic acid in a solution containing 0.085 M sodium butanoate.

Calculate the degree of ionization of 0.05 M acetic acid if its PKa value is 4.74. How is the degree of dissociation affected when its solution also contains (a) 0.01M (b) 0.1M
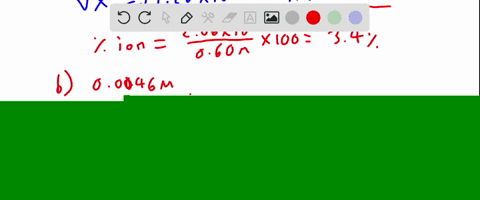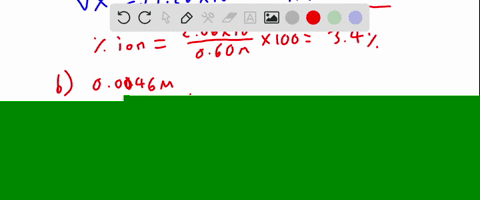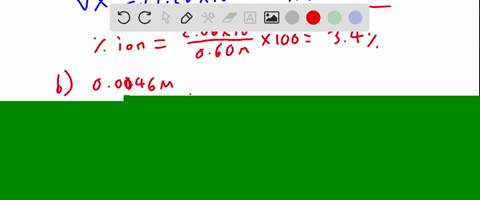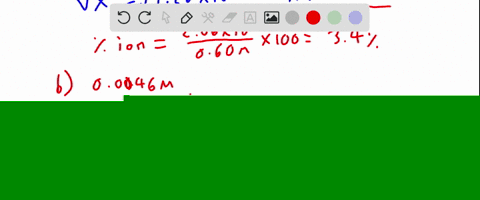
SOLVED:Calculate the percent ionization of hydrofluoric acid at these concentrations: (a) 0.60 M, (b) 0.080 M (c) 0.0046 M, (d) 0.00028 M . Comment on the trends.

SOLVED:(a) Calculate the percent ionization of 0.125 M lactic acid (Ka=1.4 ×10^-4). (b) Calculate the percent ionization of 0.125 M lactic acid in a solution containing 0.0075 M sodium lactate.
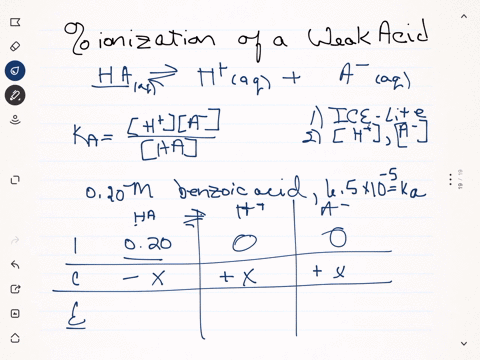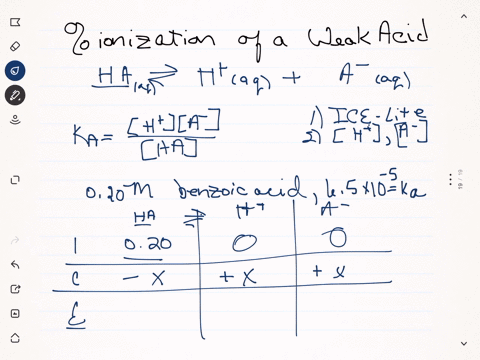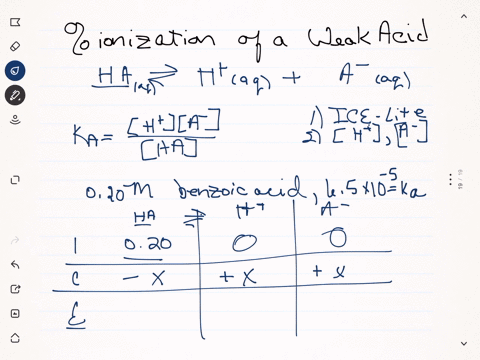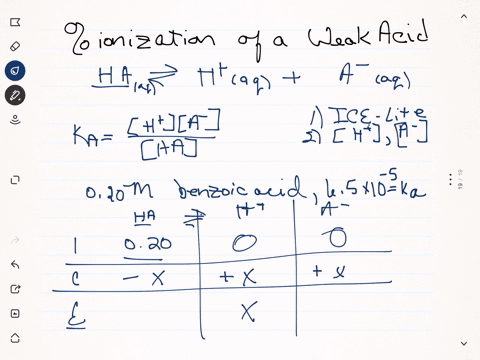
SOLVED:Calculate the percent ionization of benzoic acid having the following concentrations: (a) 0.20 M, (b) 0.00020 M.