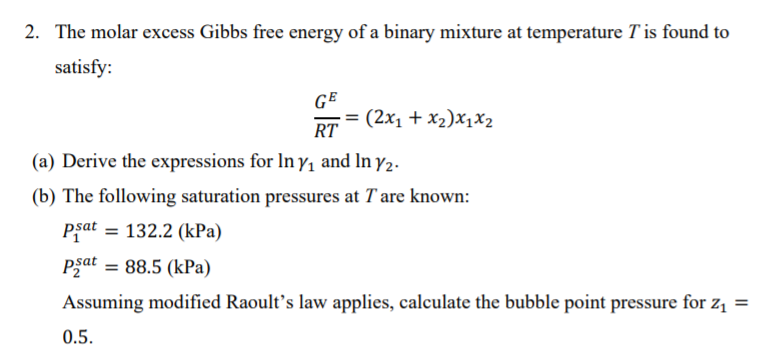27. A metal chloride dissolve endothermic , if 7.45 g of its anhydrous form dissolve in excess of of water the amount of heat absorbed is X KJ. Calculate enthalpy of solution

Fluorine reacts with uranium to form UF6 . U(s) + 3F2(g) → UF6(g) How many fluorine molecules are required to produce 2 mg of UF6 from an excess of uranium? The molar

Given the equation: Cu + AgNO 3 → Ag + CuNO 3 , what mass of silver in grams is precipitated when 40.0 g of copper reacts with an excess of silver nitrate in solution?

SOLVED: Calculate the mass of water produced when 9.47 8 of u methane CHa reacts with an excess of oxygen in the following reaction: CH4 + 202 COz + 2Hz0 HzO molar
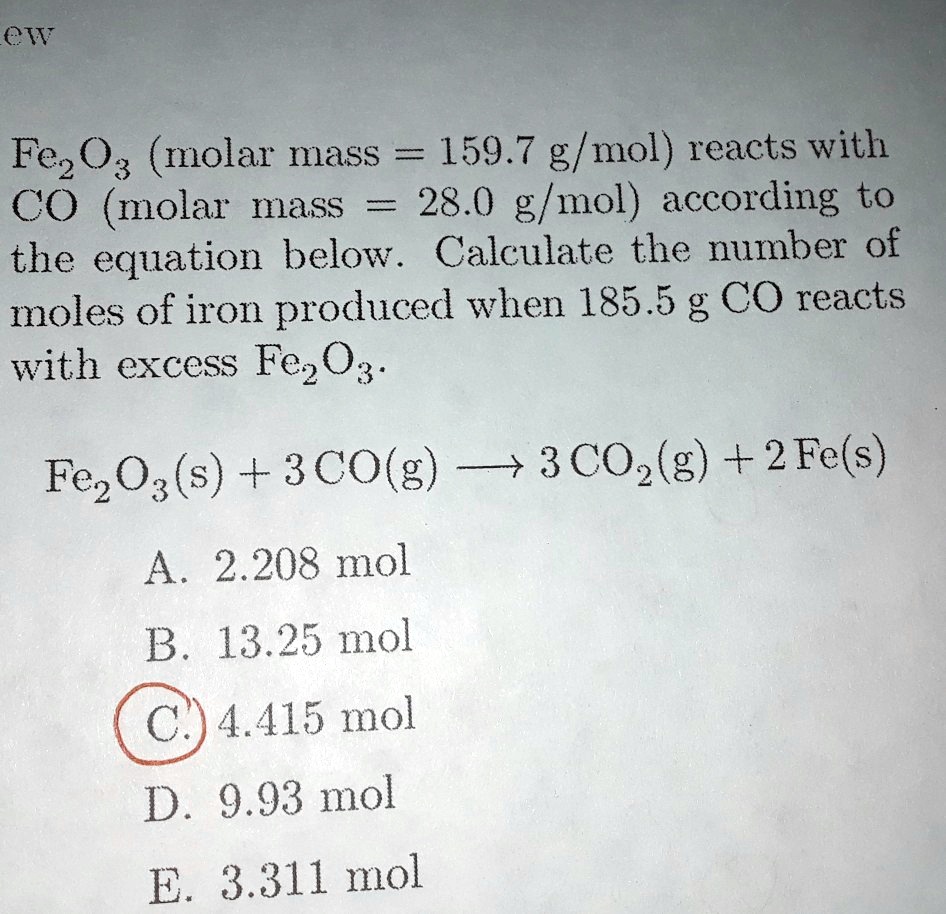
SOLVED: ew Fez O3 (molar mass = 159.7 g/mol) reacts with CO molar mass 28.0 g/mol) according to the equation below. Calculate the number of moles of iron produced when 185.5 g
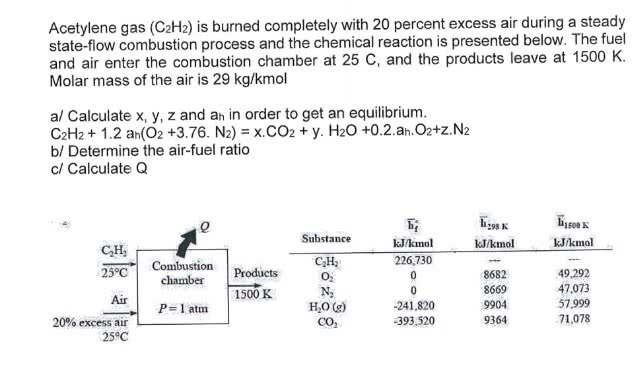
OneClass: Acetylene gas (C2H2) is burned completely with 20 percent excess air during a steady state-...