Nonproportional Hazards for Time-to-Event Outcomes in Clinical Trials: JACC Review Topic of the Week - ScienceDirect
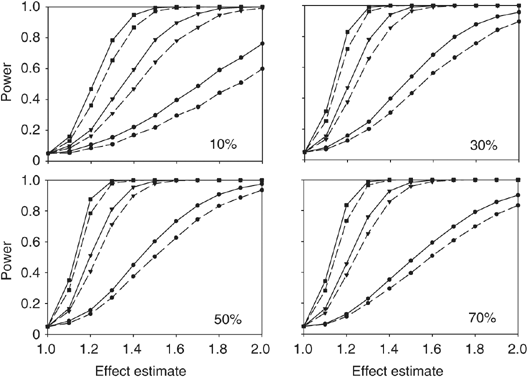
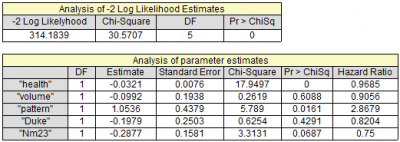
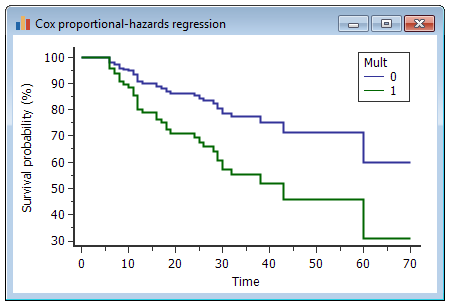
Cox proportional hazards models have more statistical power than logistic regression models in cross-sectional genetic association studies | European Journal of Human Genetics

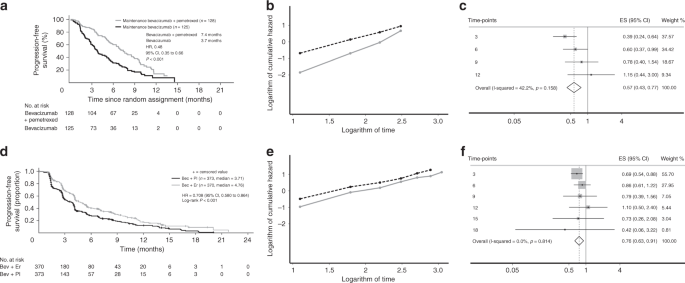
Nonproportional Hazards for Time-to-Event Outcomes in Clinical Trials: JACC Review Topic of the Week - ScienceDirect
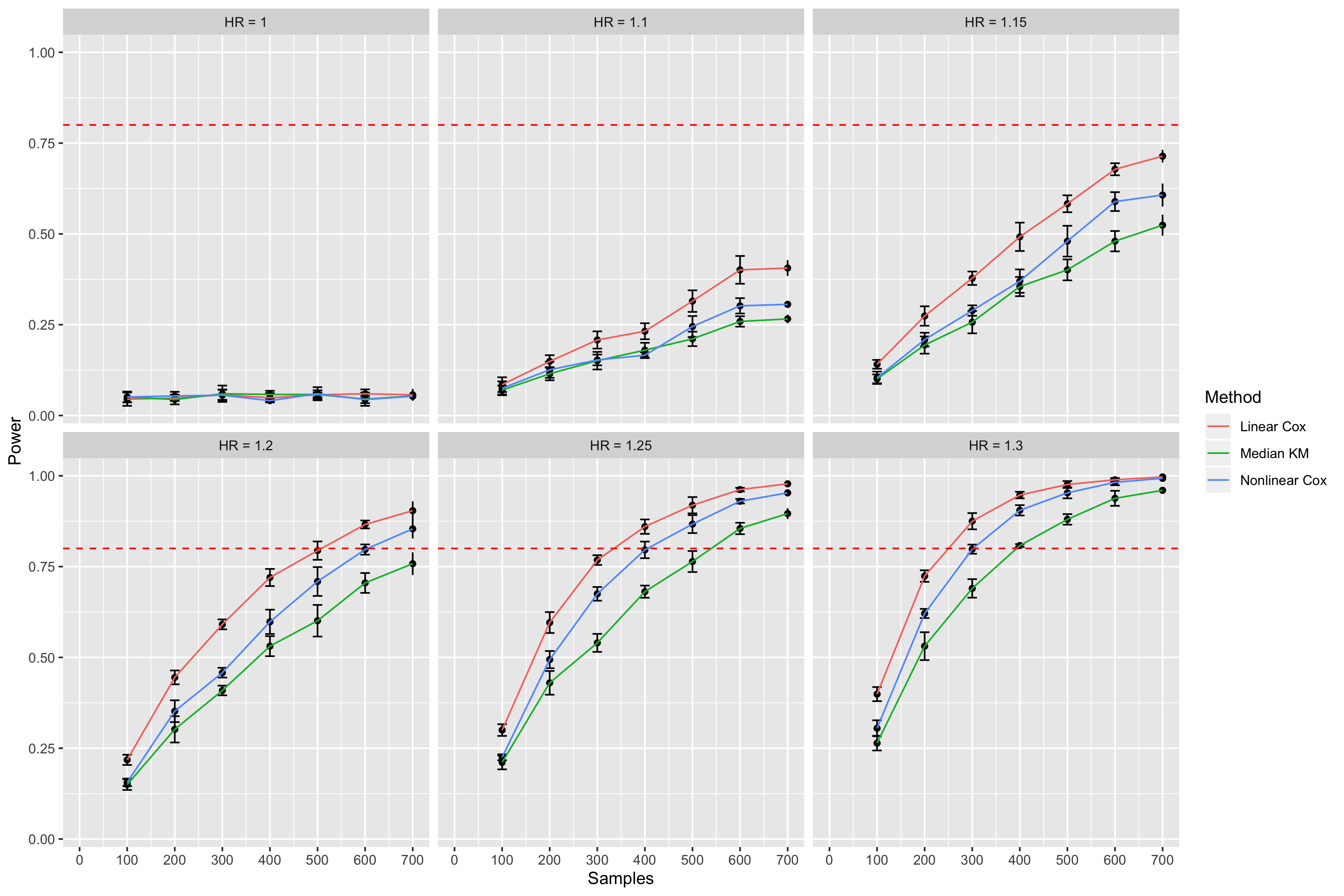
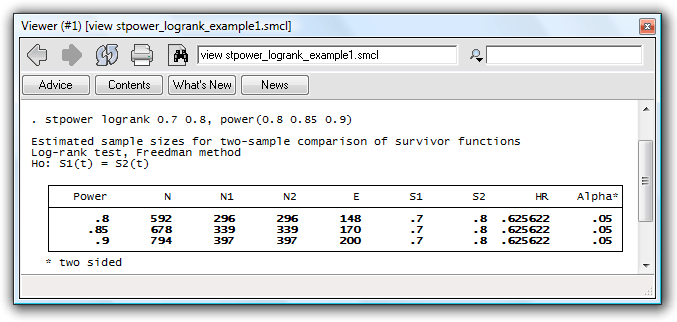
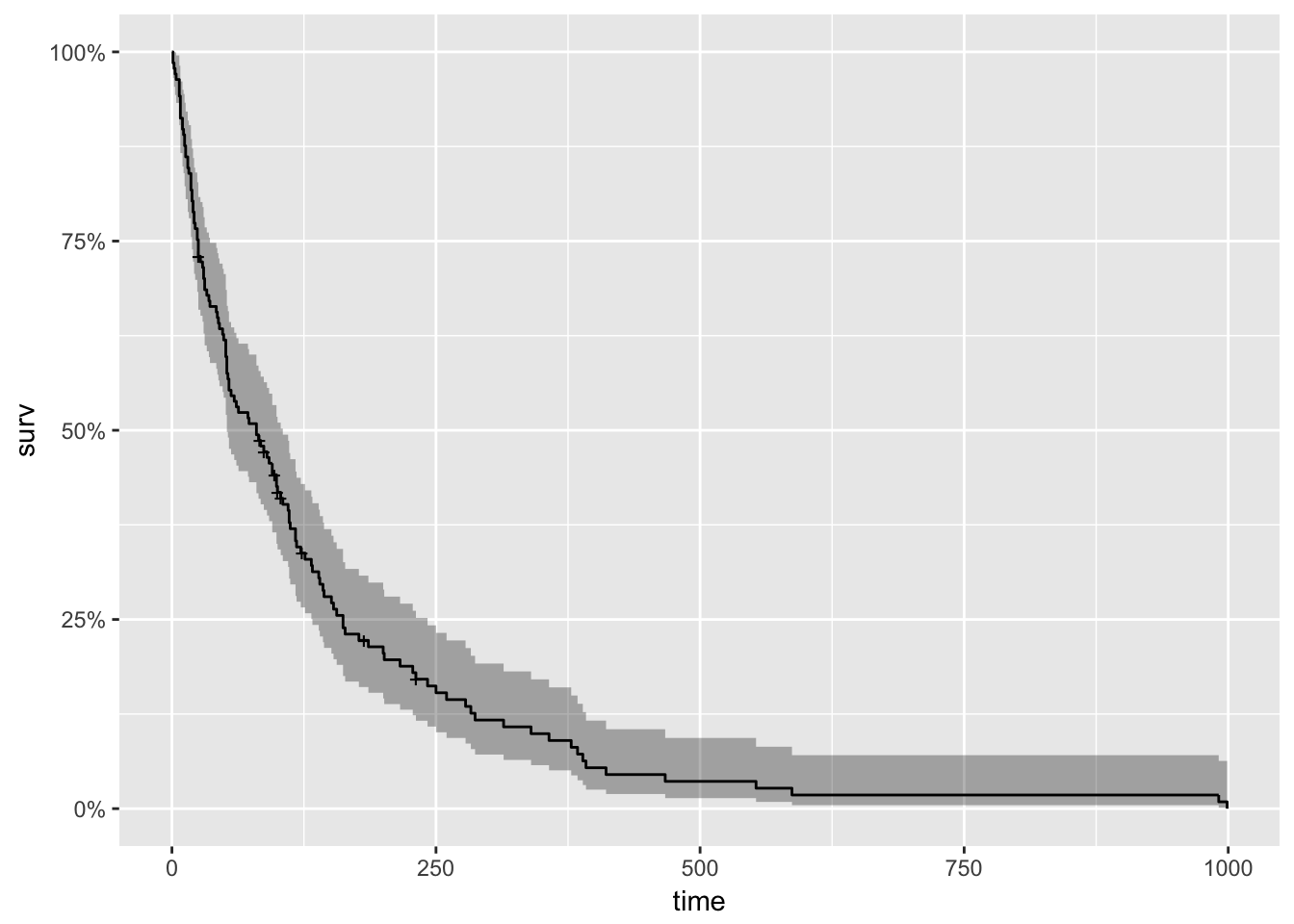
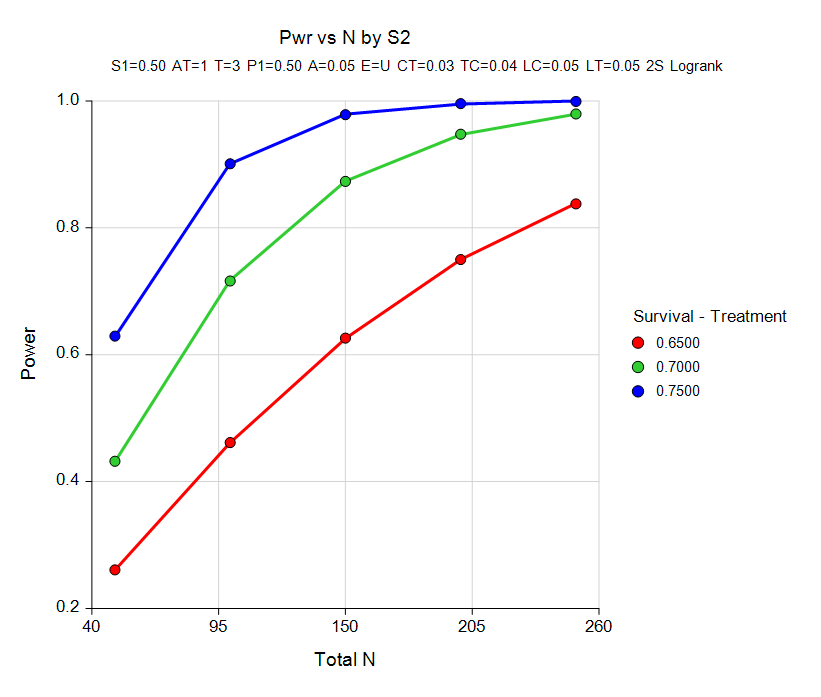
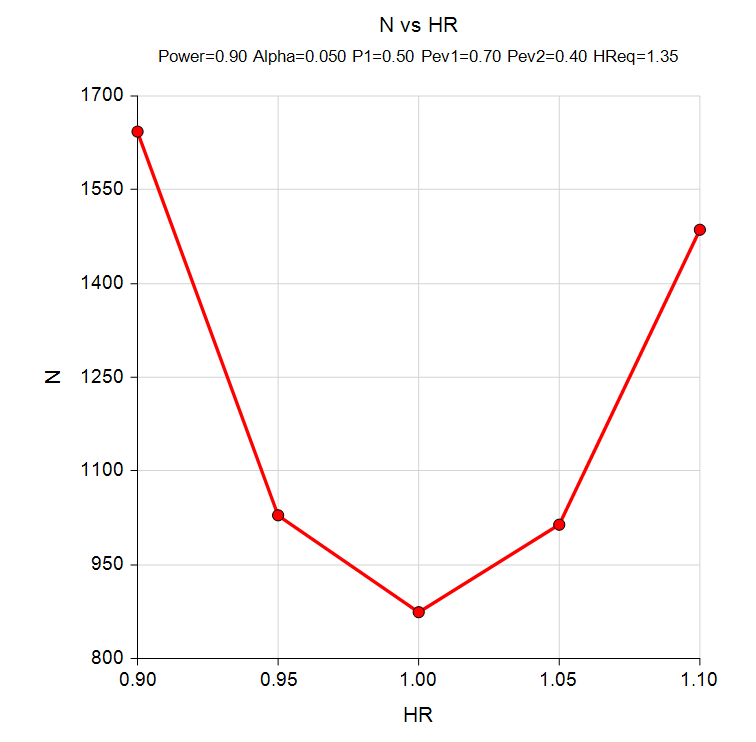
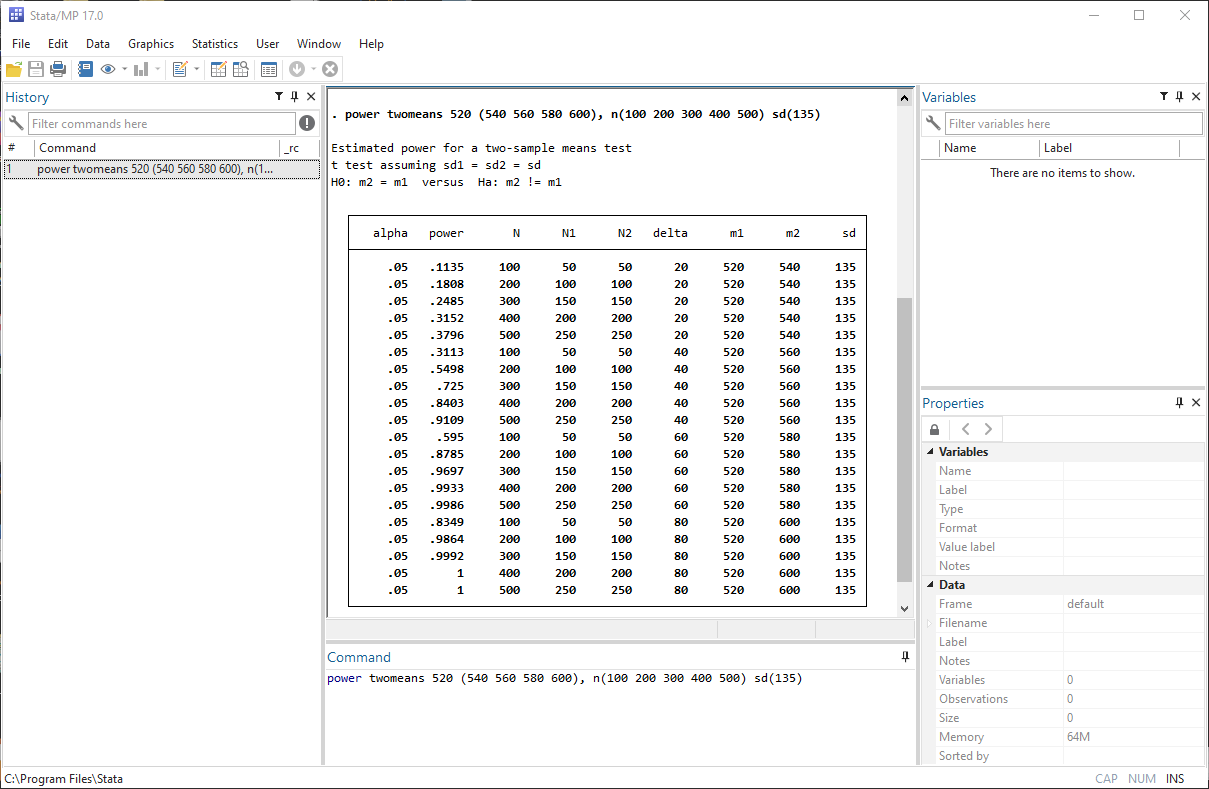
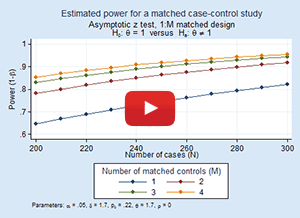
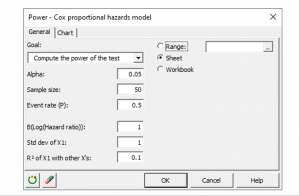
Simulating survival data to evaluate statistical power for survival analysis - modeling strategy - Datamethods Discussion Forum