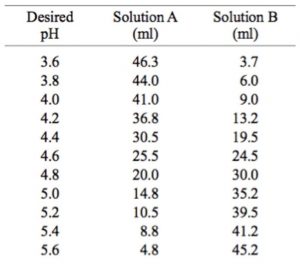
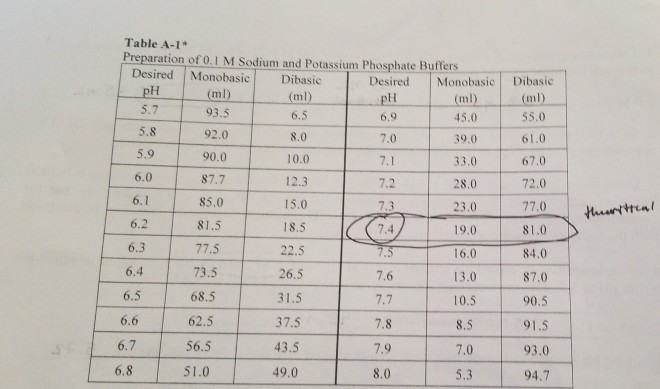
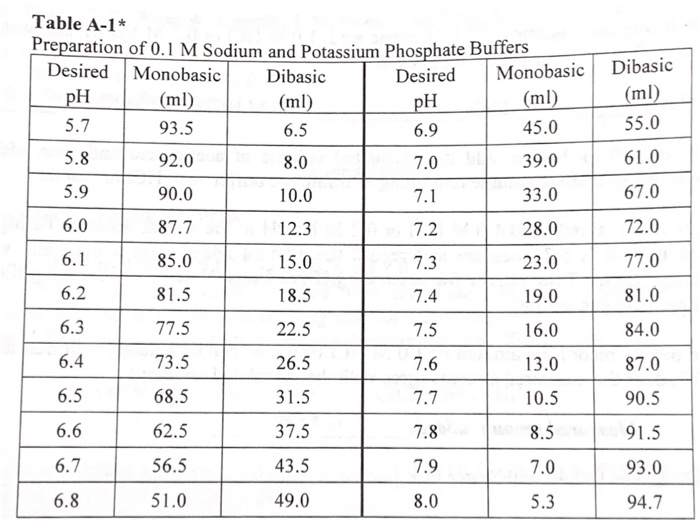
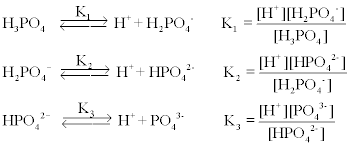SOLVED: 8 Consider beaker containing equal amounts of dihydrogen potassium phosphate (monobasic) and potassium hydrogen phosphate (dibasic) to which either a strong acid or strong base is added:. Calculate the pH of

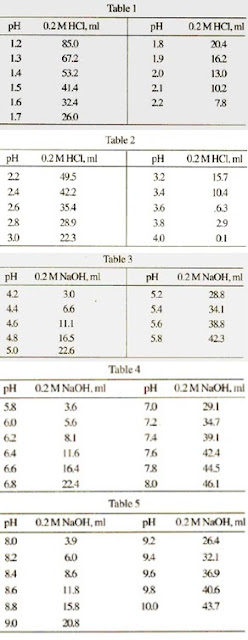
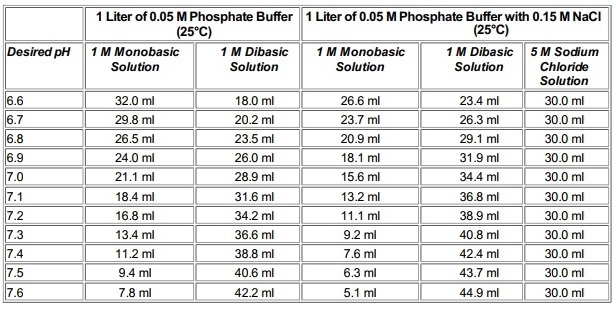
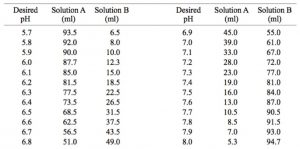
Table 1 from Dynamic approach to predict pH profiles of biologically relevant buffers | Semantic Scholar
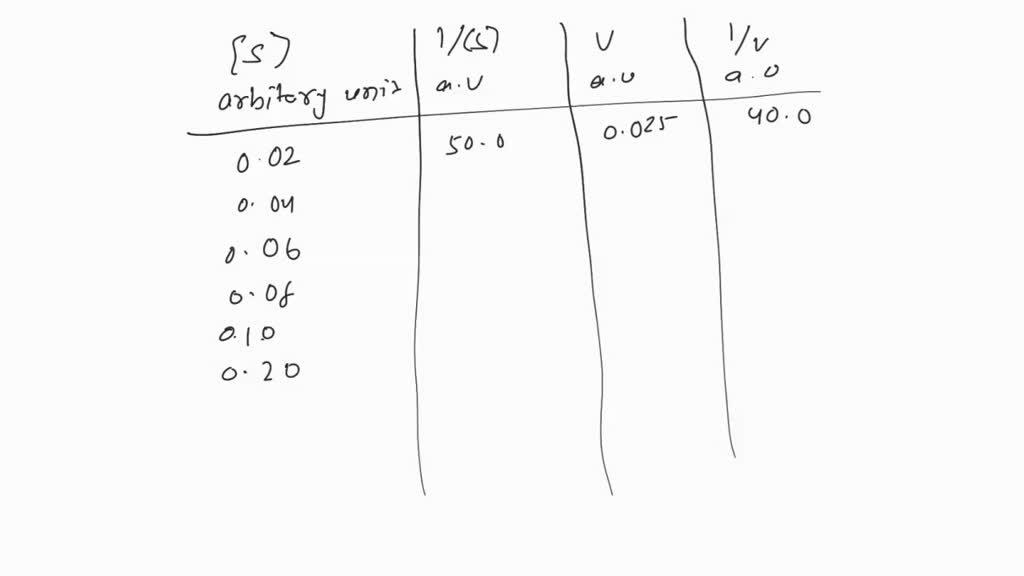
SOLVED: 1.Prepare 100 µL of 0 unit/ml and 0.5 unit/ml β-gal solution in potassium phosphate buffer. Calculate how much stock enzyme is required to prepare the diluted enzyme solution. 2.Prepare 2 tubes

Revisiting Background Signals and the Electrochemical Windows of Au, Pt, and GC Electrodes in Biological Buffers | ACS Applied Energy Materials
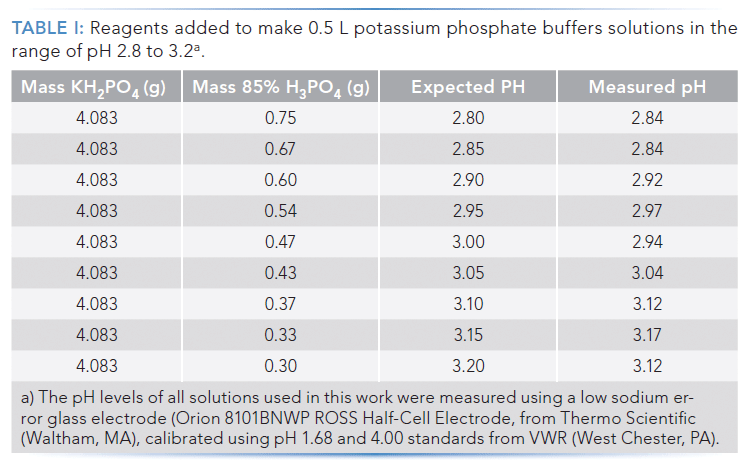
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability